Simplified PGT Workflow on a Single Embryo Biopsy

Bioarray is an innovative diagnostic and research laboratory located in Alicante, Spain.
There, scientists specialize in human genetic analysis using a variety of molecular techniques such as microarray and next- generation sequencing (NGS) technologies, including gene panels as well as whole-exome sequencing. Bioarray offers a broad menu of testing options, from clinical and reproductive genetics to research services, with expertise in preimplantation genetic screening(PGS) as well as bioinformatics.
Luis Alcaraz, PhD, is the scientific and laboratory director of Bioarray as well as its cofounder. He is a researcher in the field of proteomics and genomics, with comprehensive experience in bioinformatics using high-throughput techniques.
Thermo Fisher Scientific: You recently made some significant changes to the platform you use for preimplantation genetic screening (PGS).Can you please tell us what those changes are, and why you made them?
Alcaraz: For PGT-A, we found the scalability of the Ion ReproSeq™ PGS workflow to be advantageous to our laboratory, accommodating various sample throughputs, low or high. Therefore, as our laboratory sample throughput increases, our Ion Torrent platform grows with us; we simply purchase a different chip to accommodate higher sample throughput. The time-to-results and total hands-on time are also incredibly important to our laboratory; with the Ion ReproSeq kits, we can go from cells to data in 10–13 hours with only 2–3 hours of hands-on time, depending on sample throughput.
PGT-M is particularly challenging because traditionally it’s been very expensive, it’s difficult to design STR primers for specific disorders, and it’s not easy to combine with PGT-A. PGT-M requires a lot of expertise in primer design for interrogating specific STRs. The primers must be tested for every couple undergoing fertility treatment, and sometimes redesign of primers is needed—making the process quite lengthy, even up to several months long.
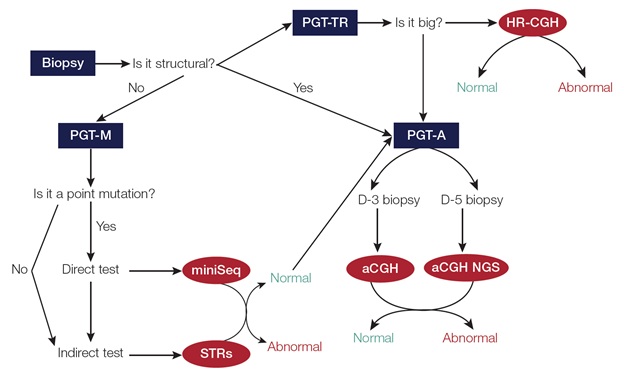
This is important because couples become stressed and do not want to wait that long. This entire process is expensive. Additionally, it’s neither easy nor cost-effective to combine PGT-A and PGT-M, because the workflows are completely different. So quite often, couples undergoing PGT-M can’t benefit from PGT-A for economic reasons.
Lastly, allele dropouts are a major concern for PGT-M, due to whole-genome amplification (WGA). For this reason, analysis of the single mutation is not enough; we need to also perform linkage analysis with several polymorphisms around the mutation.
We have been able to overcome these challenges now with Ion AmpliSeq™ technology and Ion ReproSeq™ chemistry, which work beautifully together. We can use a single biopsy for both applications, more cost-effectively. We have successfully integrated our simplified workflows for PGT-A and PGT-M in our lab with Thermo Fisher’s solutions.
Thermo Fisher: Do you have an example you can share with us to illustrate exactly how this works?
Alcaraz: Here at Bioarray, we have had tremendous success with our custom-designed kits for PGT-M based on the trusted Ion AmpliSeq technology. It is recognized globally as the gold-standard, amplicon-based enrichment method for targeted NGS. We build kits of custom NGS panels to include a 2 Mb flanking sequence on either side of the mutation locus region for linkage analysis. The PGT-M kits are designed to be used simultaneously with a single embryo biopsy required for the Ion ReproSeq workflow for PGT-A, and require only 3 µL of WGA product to start. Our unique single-nucleotide polymorphism (SNP) phasing technology and bioinformatics solution enables fast analysis of specific mutations and generates a report post–variant calling.
One common case example we see here in Europe is CFTR: I will outline the workflow for you. First, we need to perform the amplification and library preparation of the embryos. After checking the amplification, we need to take a small aliquot (about 3 µL) of the library and we use a Bioarray PGD-Seq Kit based on the Ion AmpliSeq technology to construct the library for PGT-M. Then we combine the Ion ReproSeq and PGD-Seq libraries to be loaded onto one single Ion chip for sequencing. At the same time, we need to analyze parental and relatives’ samples to perform the linkage analysis. After sequencing, we do the analysis of both PGT-A with Ion ReproSeq and PGT-M with PGD-Seq software. Finally, we combine both results to detect not only those embryos that are healthy, but also those that are euploid, increasing the effectiveness of the cycle.
Our Bioarray PGD-Seq Kits are available for virtually all single-gene disorders, including cystic fibrosis, spinal muscular atrophy, beta-thalassemia, polycystic kidney disease, fragile X syndrome, and others.
Thermo Fisher: What advice do you have for other laboratories who may be running aCGH or thinking of consolidating PGT-A and PGT-M workflows?
Alcaraz: For aneuploidy testing, aCGH is an obsolete technique. Now we know how important it is to correctly identify mosaic embryos, and thanks to the higher accuracy and sensitivity of NGS, we can categorize the embryo as normal, abnormal, or mosaic. Moreover, we can use the last category as the last chance for many couples when there are no more normal embryos.

Another important point is around PGT-M. Nowadays, thanks to the wide use of carrier screening panels, many couples know that they are at risk of having a child affected by a recessive disease. Now they have the opportunity to detect which embryos are not affected by the disease.
Thanks to NGS, integrating the PGT-M workflow with PGT-A has several advantages, including no investment needed for additional equipment. Classically, PGT-M requires a capillary sequencer, but now with the Ion AmpliSeq technology, it is possible to perform testing with the same equipment you already have for PGT-A. Both PGT-M and PGT-A can be run on the Ion GeneStudio™ S5 System. Another advantage is less sample handling is needed, thereby reducing the possibility of error. Combining both protocols is more cost-effective, allowing more couples to benefitfrom PGT-A and PGT-M.Lastly, we have the advantage of robust bioinformatic analysis for both PGT-Aand PGT-M. Our simplified PGT-A andPGT-M workflow on a single embryo biopsy has transformed the testing we can offer.
Source : thermofisher


